The goal of preclinical drug development is to identify potential therapeutic compounds and gain an understanding of disease pathways or target mechanisms. Although preclinical efficacy studies aim to mimic the clinic setting, several factors are overlooked that limit the understanding of drug efficacy. For example, preclinical efficacy studies, especially in pain, are predominantly performed in male species. However, there has been recognition of the importance of including both sexes in studies to better understand pain perception and response to treatments. When designing a preclinical study, it is crucial to consider differences not only in sex, but also in age, species, comorbidities, and genetic background. Including these factors early on increases clinical translatability and leads to more effective pain management.
In recent years, MD Biosciences has invested resources in studying the effects of diversity in preclinical studies. Our neuroscience team is building a data bank that addresses factors, like sex and age, in several pain and neurodegenerative models. In this post, we share recent data highlighting these differences.
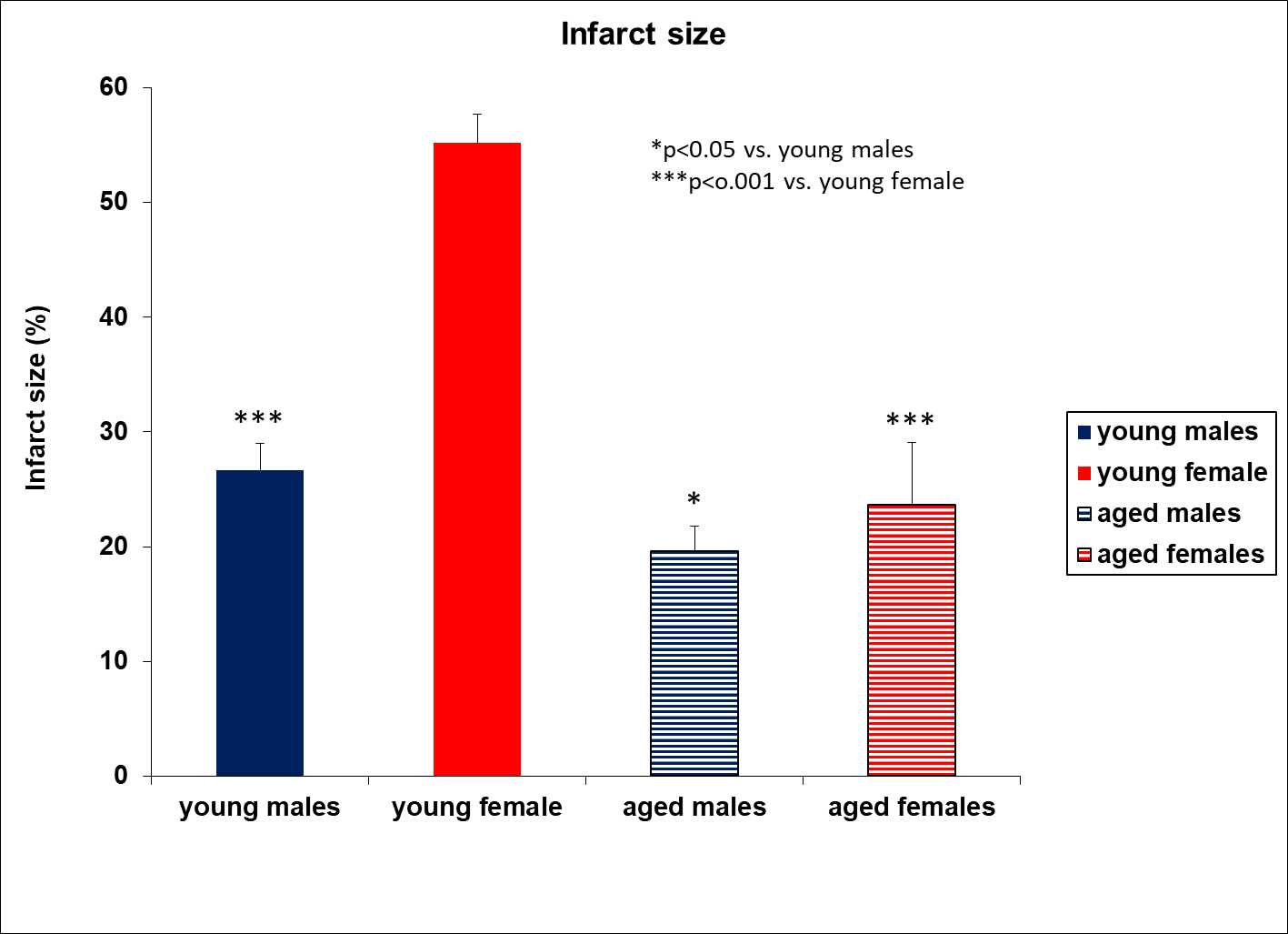
Example 1: In the MCAo model for stroke, young male animals are standardly used, which does not reflect the clinical setting. When we evaluated both sexes in young and aged animals, we observed differences in infarct size and the grip strength test.
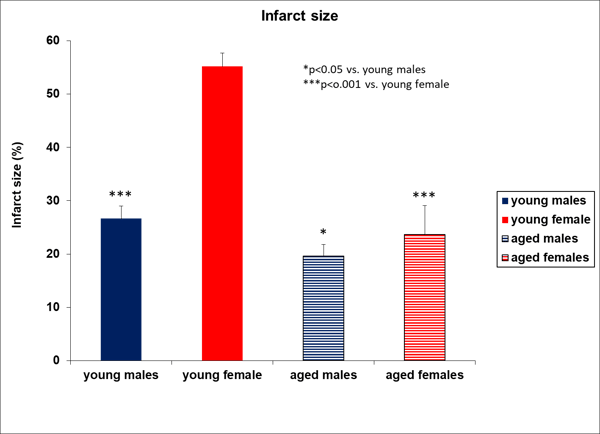
Figure 1. Aged rats express smaller infarct size compared to young males.
! data in process and will be available soon.
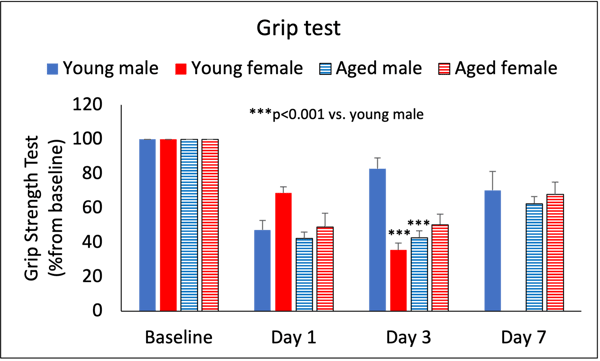
Figure 2. Grip strength test in young and aged male and female rats. On day 1, there is an expected decrease in the grip strength test. On day 3, grip strength in females decreases compared to young males. Similarly, grip strength in aged males decreases compared to young males. On day 7, all groups except for young females demonstrate ~75% recovery.
Example 2: In the STZ-induced model of neuropathic pain, we compared pain response in male and female rats.
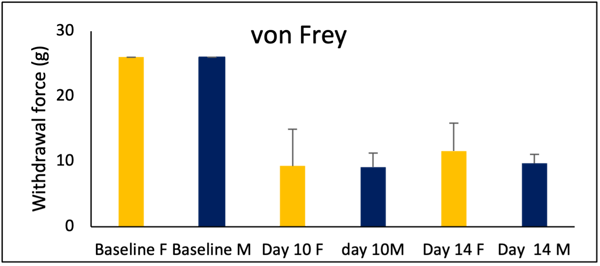
Figure 3: In the von Frey test, female and male rats show increase in sensitivity to tactile stimulation in STZ induced peripheral neuropathy.
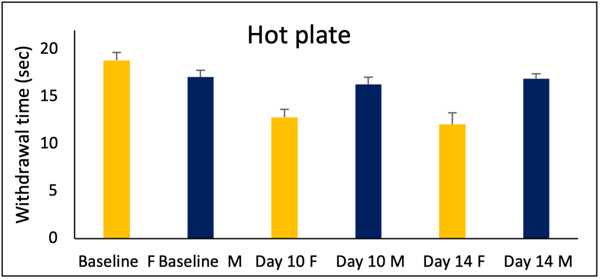
Figure 4: Only female rats show sensitivity to the hot plate test.
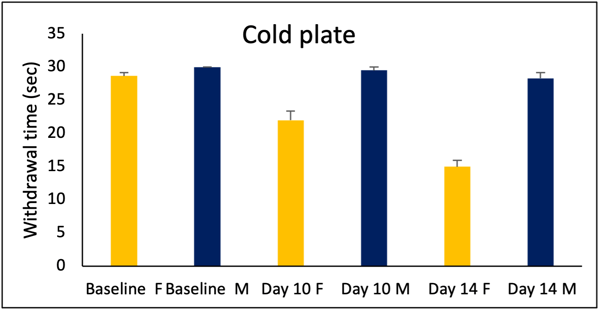
Figure 5: Only female rats show sensitivity to the cold plate test.
Designing preclinical studies with diversity in mind can provide stronger predictions of how drugs will perform in diverse human populations. As a research partner, we aim to address these variables early on to improve data quality and increase translation to the clinic. To learn more about our work, watch this webinar to understand:
- diversity considerations from tissue culture studies to large animal models.
- see examples of age-sensitivity, sex differences and comorbidities in preclinical studies.
- get a review of data that shows the discrepancy between preclinical efficacy outcomes and the clinical situation.