Cell-based Assays
Efficacy Models
PK/PD & Toxicology
Pain
Neurodegeneration
Neuroinflammation
Wound Healing
Content
Diversity in Efficacy Studies.
Introducing diversity in non-clinical and translational CNS models for better prediction of clinical outcomes.
Watch Webinar
Diversity matters.
Lack of diversity in clinical and preclinical studies creates obstacles in understanding safety and efficacy of therapies across subgroups. What considerations can be added to preclinical efficacy studies to better predict clinical outcomes?
About this webinar:
25 minutes on-demand
Late stage failures in drug development are costly and time-consuming. Much debate continues to take place over the predictability of animal efficacy models. As a nonclinical CRO focused on increasing the translatability and predictability of efficacy studies within CNS and pain, we address the following in this webinar:
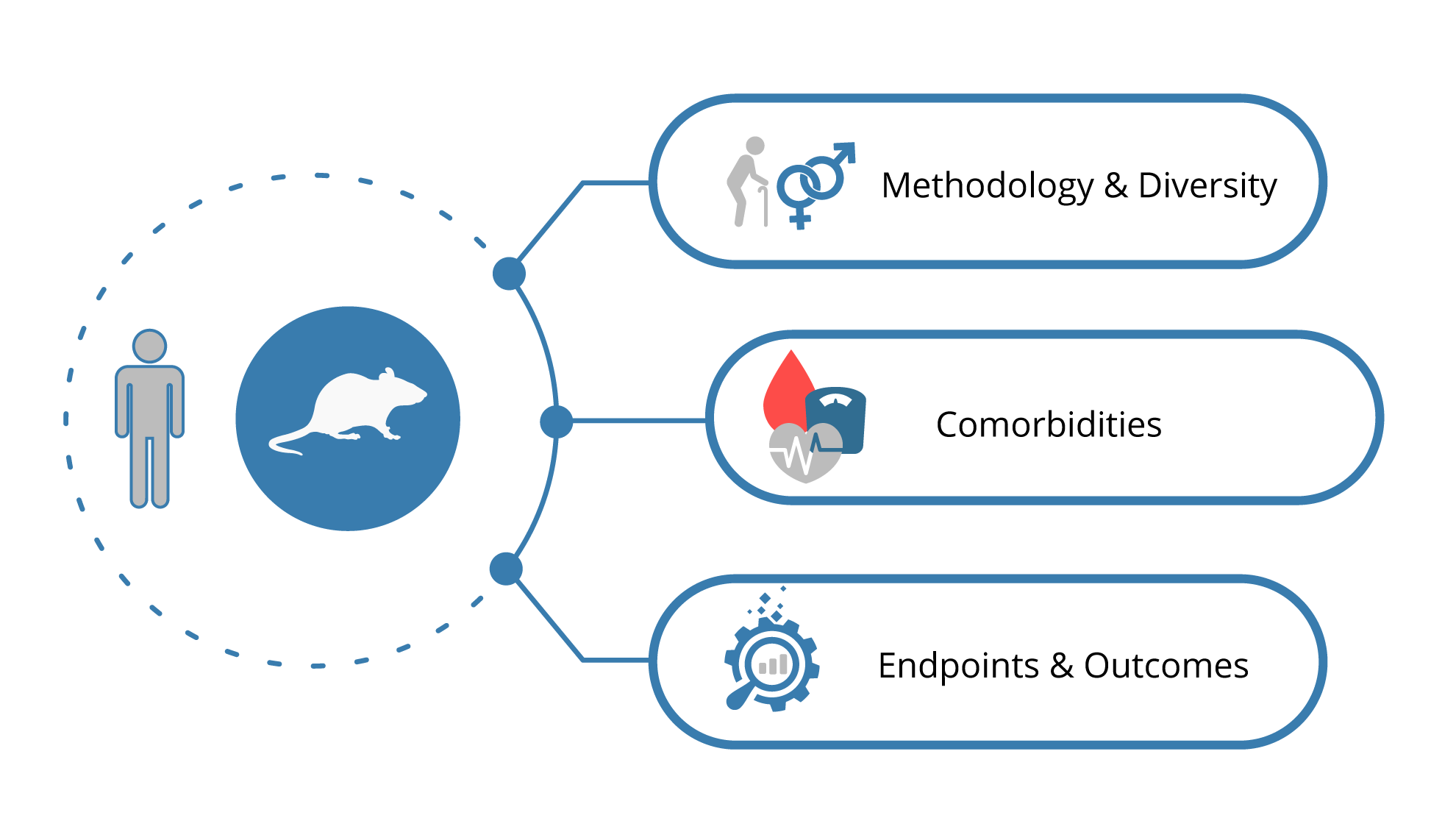
- Diversity considerations from tissue culture studies to large animal studies.
- Examples of age-sensitivity, sex differences and comorbidities in preclinical studies and the discrepancy to the clinical situation that is created.
- Data to show the discrepancy between preclinical efficacy outcomes and the clinical situation.
"Diversity should be taken into account all the way from in vitro studies to large animal efficacy studies."
Dr. Sigal Meilin
Chief Scientific Officer




