Remitting relapsing PLP-induced EAE Efficacy Model for MS Research
Proteolipid Protein (PLP) is a major protein component of CNS myelin. Sections of PLP, such as peptide fragment 139-151, are encephalitogenic to certain mouse strains. PLP injected in Swiss Jim Lambert (SJL) mice together with pertussis toxin (PT) will lead to remitting and relapsing demyelinating disease. Relapses are associated with epitope spreading, in which T cells react to secondary endogenous peptides that emerge as a consequence of the initial phase of myelin destruction. Remissions are associated with a temporary loss of inflammatory cells from the CNS.
-
Characteristics of PLP-EAE
In our Multiple Sclerosis (MS) efficacy model, disease is induced on day 0 using PLP emulsified with Complete Freud's Adjuvant (CFA). Additionally, to increase the susceptibility of the Blood Brain Barrier (BBB), all animals are subject to supplemental immunostimulation with a PT injection on day 0 and day 2. The disease typically develops between days 10–12 with the peak occurring between days 14-16. If the relapse phase is required, the study can be extended such that a second disease peak will be apparent between days 25-30. This disease is characterized by paralysis attacks of all 4 paws and is typically used for testing the activity of a compound by injection of the compound between phases.
-
Assessments
- Clinical Score
- Histology for cellular infiltration
- Immunohistochemistry (IHC) for demyelination
- Biomarkers
- PK
Assessments
MD Biosciences incorporates a range of in vivo measures and endpoint assessments that provide a robust data package that enable researchers to make critical decisions.
Behavior
Pain behavior tests include dynamic weightbearing and Hargreaves to assess the distribution on each paw and sensitivity to heat.
Biomarkers
Evaluate pro-inflammatory and anti-inflammatory biomarkers from CSF samples.
Histology
IHC and histological staining to evaluate inflammatory markers in the paw skin.
Scientific Data
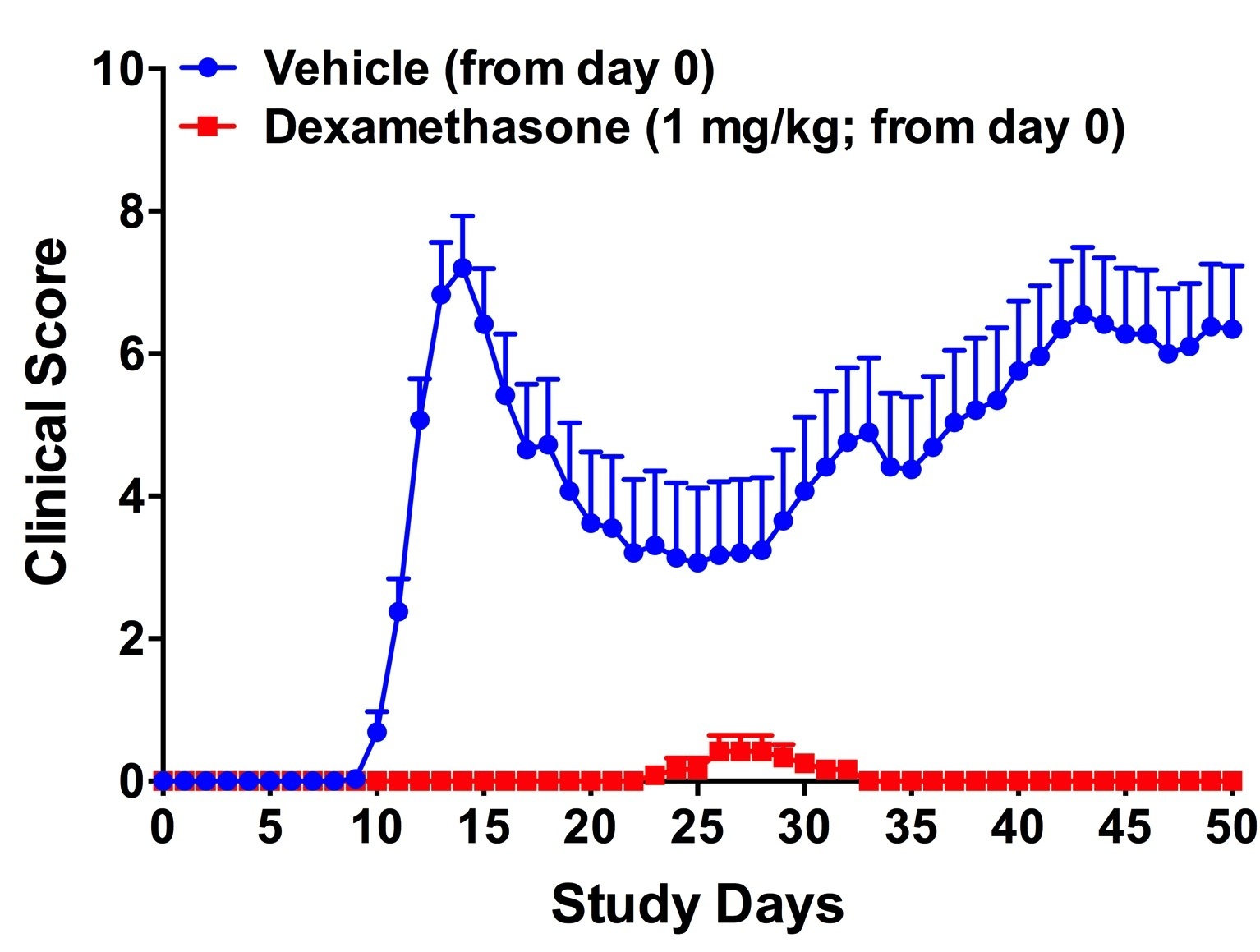
Clinical score in the PLP-induced EAE model indicating the peaks of disease. The first phase of the disease begins around day 10 and subsides around day 25. On day 40 the relapse begins.
Review the complete dataset.
![]()
EAE Datasheet
Complete the short form for instant access to the complete data set.



